COMSOL has the best multiphysical simulation capabilities in my experience. Technical support from Elisa at TECHNIC as well as the engineers at COMSOL has been great.
COMSOL is an important part of our research in plasma physics. We use it in the design of plasma systems and it helps us to obtain a greater understanding of the underlying physics. We have always valued the quick support from TECHNIC and COMSOL and it has been a pleasure to work with them.
Comsol has become a valuable part of our design and decision making process. The exceptional flexibility and access to the physics and solvers in Comsol has allowed us to have deeper understanding on thermomechanical solutions. Technic and Comsol have always been quick and helpful to resolve any issues and provide helpful advice on their products.
At Scion we use COMSOL Multiphysics to understand energy processes, such as the interplay of non-linear solid mechanics and heat & mass transfer during biomass compaction, to design new or more efficient processes.
We use COMSOL Multiphysics to design the customised muffler. With it, we can simulate the insertion loss at different spectrum with different muffler designs.
Modelling and simulations are cost-effective methods for understanding, optimising, and controlling electrodeposition processes. A typical simulation yields the current distribution at the surface of the electrodes, and the thickness and composition of the deposited layer. Simulations are used for studying important parameters such as cell geometry, electrolyte composition, electrode reaction kinetics, operating voltages and currents, as well as temperature effects. With information about these parameters, you can optimise the operating conditions of the electrochemical cells and the placement and design of masks, and ensure the quality of your surfaces, while minimising material and energy losses.
Decorative electroplating assuming secondary current distribution with full Butler-Volmer kinetics for both the anode and the cathode. The deposited thickness on the front and the backside of the piece is shown.
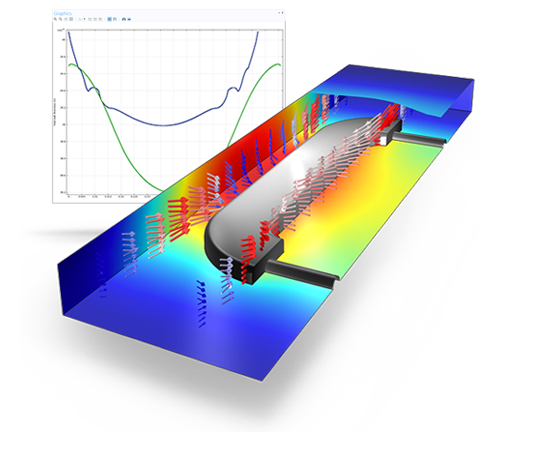
The Electrodeposition Module is suited for a wide variety of applications, including: metal deposition for electronics and electrical parts; corrosion and wear protection; decorative electroplating; electroforming of parts with thin and complex structures; etching; electromachining; electrowinning; and electrorefining. With the Electrodeposition Module, you can consider all of the participating phenomena and simulate them together. More specifically, you may couple the equations that describe current transport and conservation, chemical species transport, charge balances, and electrochemical kinetics. Due to the ability to account for several relevant phenomena, you are able to obtain accurate estimates of the quality, shape, and thickness of the deposit on the surface of the electrodes.
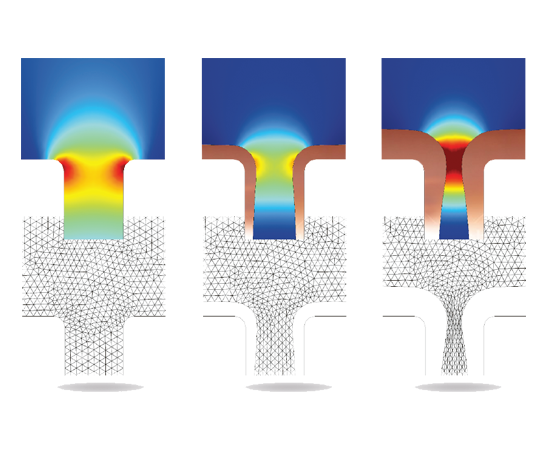
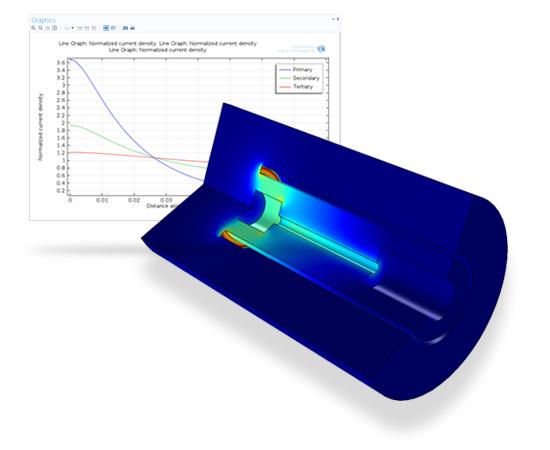
Tools and physics interfaces are available within the Electrodeposition Module for defining the physical characteristics of your process. Predefined formulations allow you to model primary, secondary, and tertiary current distribution effects – often excellent indicators of the surface finish and product quality of your process.
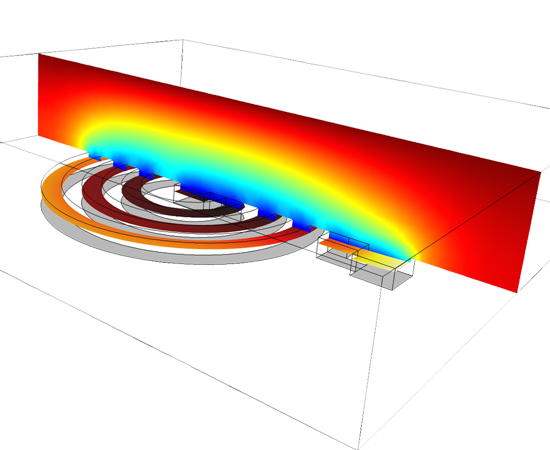
As with all chemical processes, you can study many of the effects of your electrodeposition processes at different scales, depending on your purpose. On the microscale level, this could mean investigating the electrochemical kinetics of the participating reactions and the influence of different levellers or striking conditions on the kinetics of electrodeposition. At the counter electrodes, a careful selection of electrocatalyst and electrode micro structure may be required, especially for electrowinning where you need to minimise losses. Simulating these processes and comparing them to experiments or data from your process will allow you to understand the charge transfer reaction mechanism at the electrode surfaces, and derive the electrochemical kinetics parameters, like the exchange current densities and the charge transfer coefficients for each reaction. By knowing these mechanisms and parameters, you can simulate the rate and distribution of deposition or etching at a larger scale. Simultaneously, you will be accounting for the operating conditions of the system, such as cell and electrode geometry, cell voltage or imposed current, masking and shielding, electrolyte composition and flow, gas evolution, and temperature.
The standardised COMSOL Desktop® user interface, which is the basis for the Electrodeposition Module, is also used for all the other add-on products in the COMSOL Product Suite. This enables you to couple the physics describing your electrodeposition or etching process to other modules, such as the Heat Transfer Module for studying thermal effects, or the CFD Module for understanding the effects of two-phase flow. Moreover, other physical characteristics, like structural integrity for example, can be modelled sharing the same model files that you used to model your electrochemical cell. This unified modelling platform is the perfect tool for collaborating with engineers working on the same process across different fields.
The Electrodeposition Module comes with a number of easy-to-use tools for many of the characteristics associated with electrodeposition and electroplating cells. These include the following:
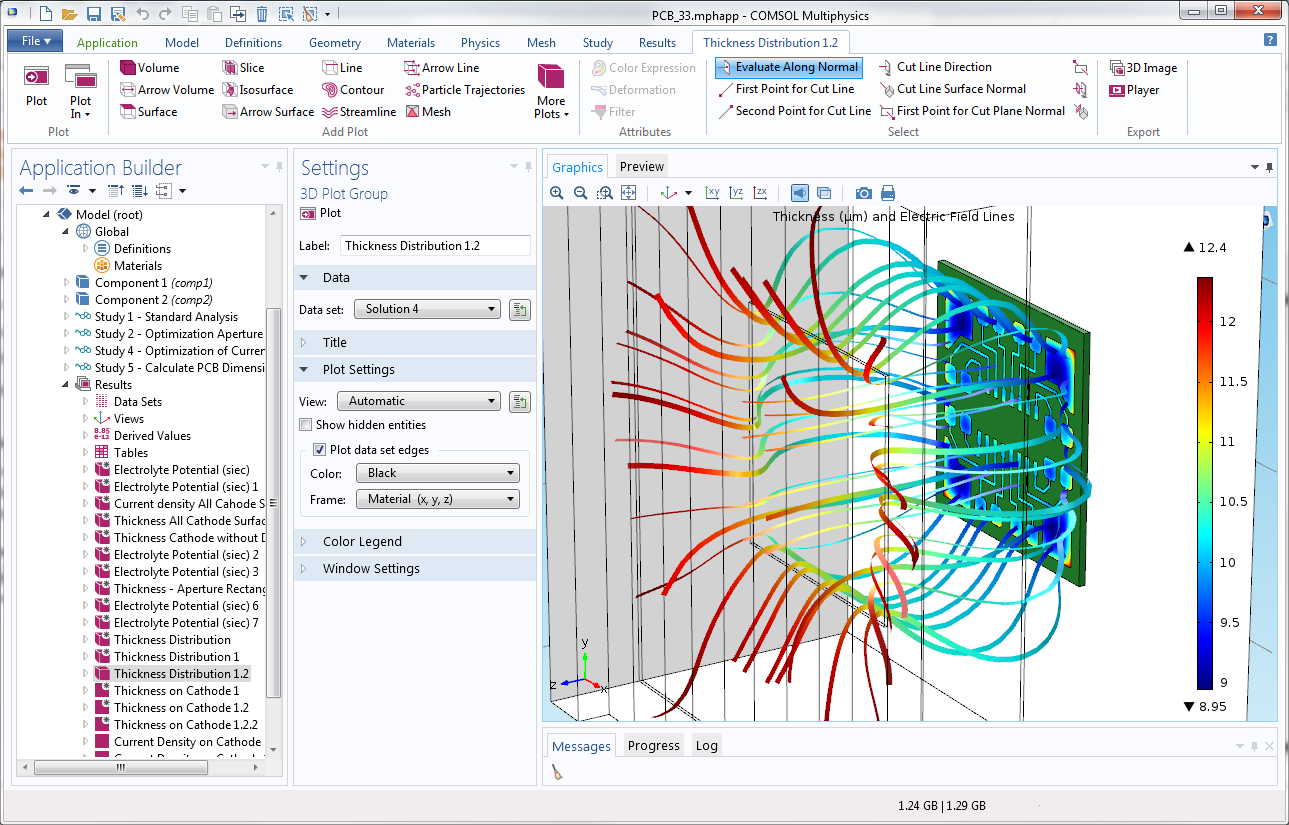
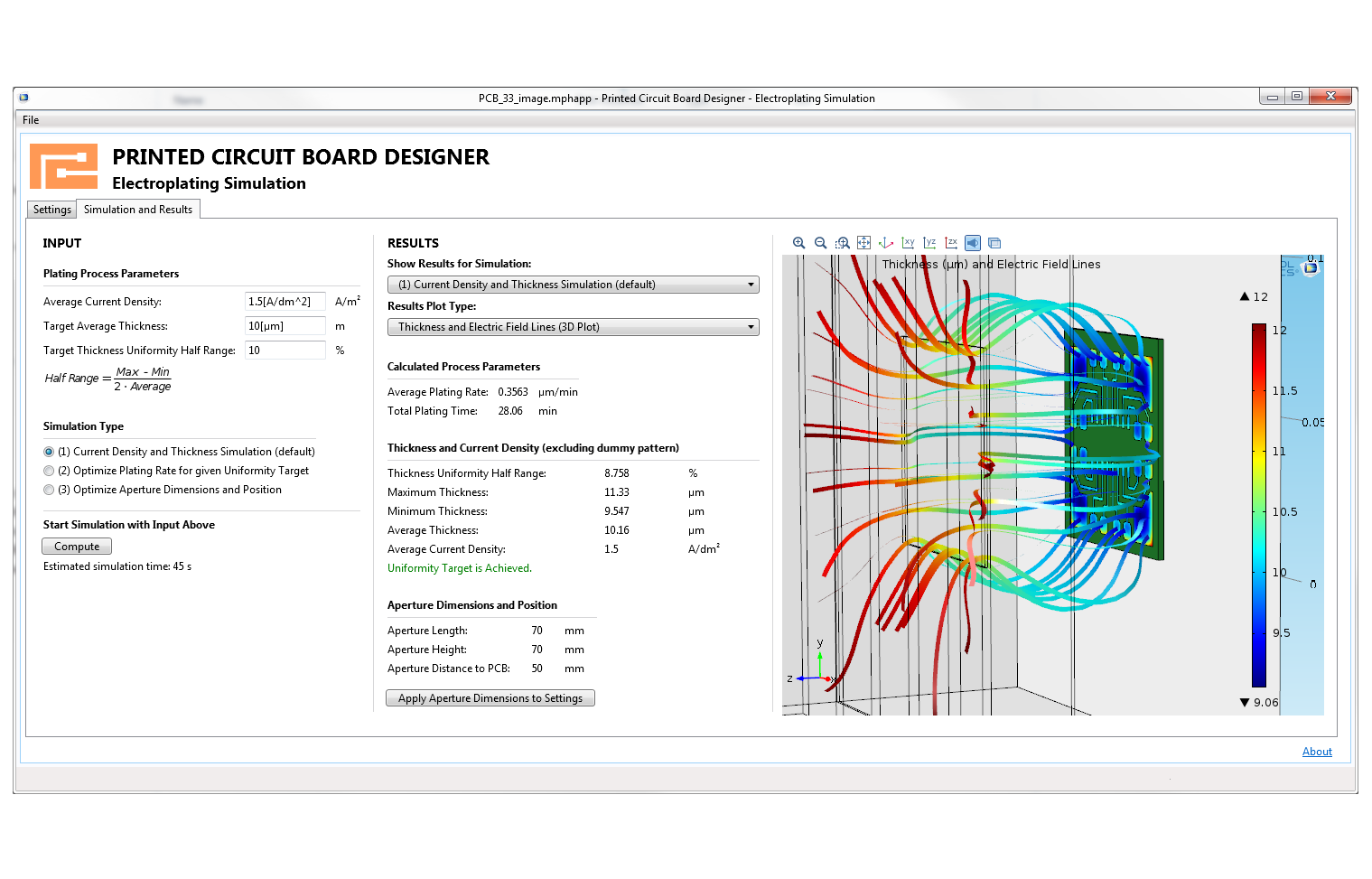
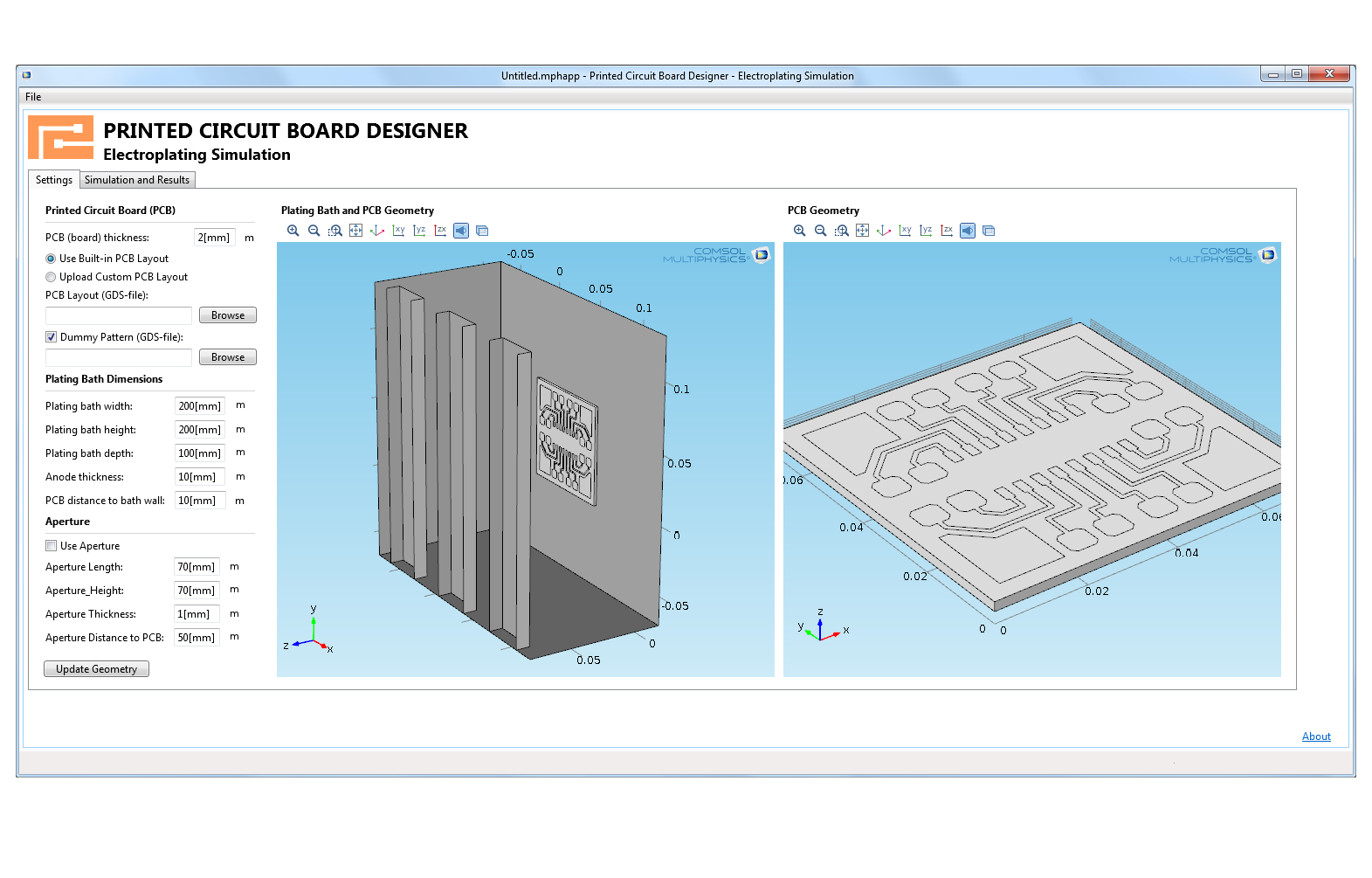
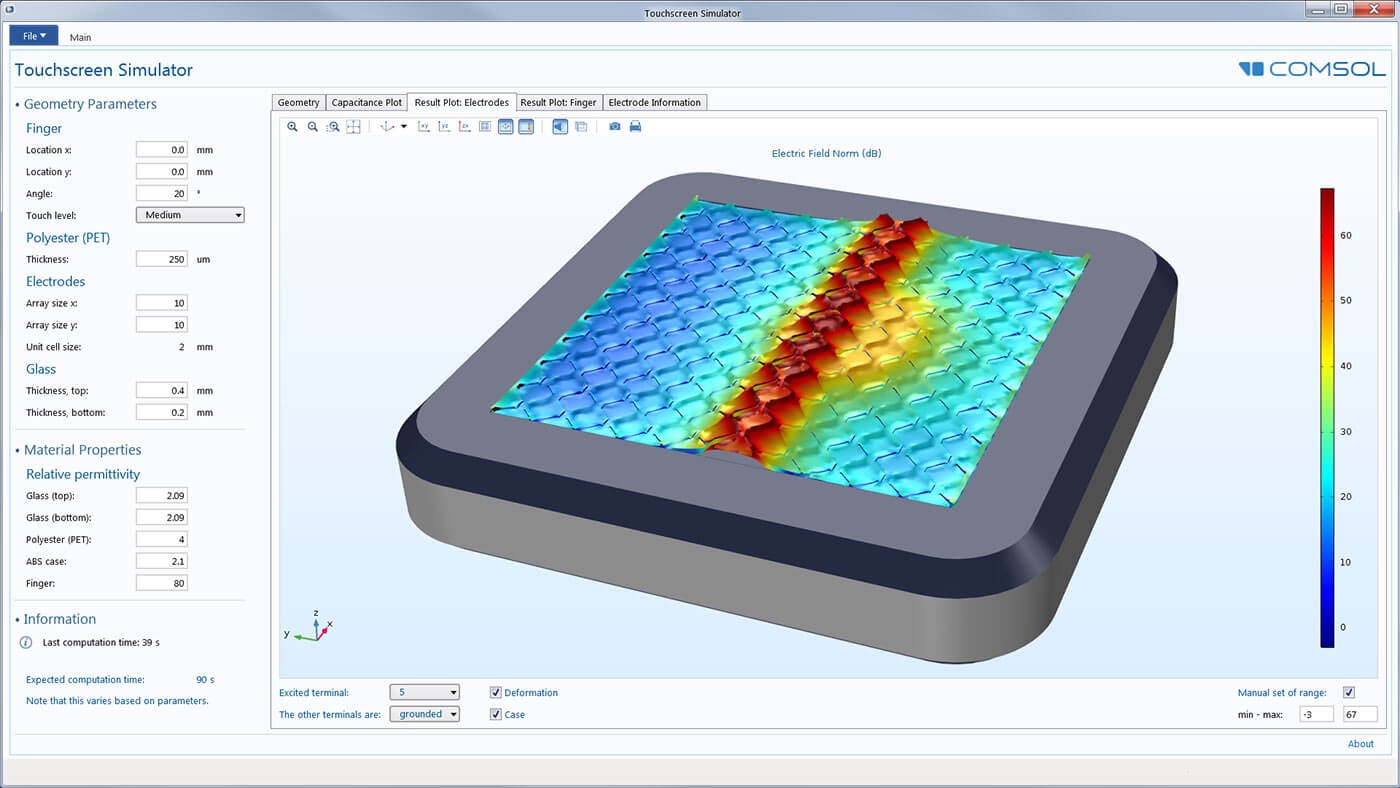
Make your simulation process more efficient with the built-in Application Builder by turning your models into specialised applications with customised inputs and outputs. You can distribute the applications you make to colleagues without simulation expertise so they can perform repetitive analyses on their own, streamlining the design process.
The workflow is simple:
You can expand the capabilities of simulation throughout your team, organisation, classroom, or customer/vendor base by building and using simulation apps.
If you use the MATLAB® software, you can easily drive COMSOL Multiphysics® simulations with MATLAB® scripts and functions. The LiveLink™ for MATLAB® interfacing product enables you to access COMSOL® operations through MATLAB® commands and blend these commands with your existing MATLAB® code, directly within the MATLAB® environment.
In order to make it easy for you to analyse electromagnetic properties of CAD models and electronic layouts, COMSOL offers the ECAD Import Module, CAD Import Module, Design Module, and LiveLink™ products for leading CAD systems as part of our product suite. The LiveLink™ products make it possible to keep the parametric CAD model intact in its native environment but still control the geometric dimensions from within the COMSOL Multiphysics® software, as well as produce simultaneous parametric sweeps over several model parameters.
You can also synchronise Microsoft® Excel® spreadsheet data with the parameters you define in the COMSOL Multiphysics® environment via the LiveLink™ for Excel® interfacing product.
View all available interfacing products in the product suite.

In order to fully evaluate whether or not the COMSOL Multiphysics® software will meet your requirements, you need to contact us. By talking to one of our sales representatives, you will get personalised recommendations and fully documented examples to help you get the most out of your evaluation and guide you to choose the best license option to suit your needs.
Fill in your contact details and any specific comments or questions, and submit. You will receive a response from a sales representative within one business day.